 Alkene Reactions Series: Video 1
Alkene Reactions Series: Video 1
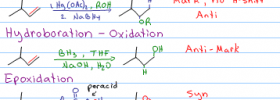
This introduction to organic chemistry alkene reactions tutorial is meant to serve as an introduction to the concept of alkene reaction mechanisms. Starting from the very foundation, learn what makes alkene electrons so reactive, as well as how to recognize a nucleophile and electrophile in orgo reactions.
You'll also get a quick overview of alkene reaction mechanisms and carbocation intermediates.
(Watch on YouTube: Intro. Click cc on the bottom right for video transcription.)
–> Watch Next Video: Hydrohalogenation of Alkenes
This is Video 1 in the Alkene Reaction Mechanisms Video Series. Click HERE for the entire series.
Ready to test your skills? Try the Alkene Reactions Practice Quiz after watching the series!


Really helping me to understanding my whole chemistry and organic chemistry which i really like to study but couldnot figureout properly, videos tutorials are great and cheat sheet are really awesome. Thank you leah. Really glad to find you. PRATIK (M.sc chemistry students) FROM Nepal
Leah, isn’t this the first video of the series?
Leah, isn’t this the first video of this series ?
why doesn’t pi top and pi bottom make 2 pi-bonds instead of 1? and how does the p-orbitals form a bond if they don’t face each other?
let alone make the pi bond more electron-rich somehow
You’re amazing! Thanks for your help and sharing knowledge in such a simple and easy to understand way 🙂
You’re very welcome Cheryl. Glad to help
In this type of alkene reactivity we need to follow Markovnikov rule for substitution
For which reaction?
Leah, thanks for making these great videos!
As I was watching this video, something that I had to pause and really think about was that you would describe the 2-butene’s double bond as “electron rich” (and therefore nucleophilic) especially since there valence electrons were tied up making the pi bond. The octet was satisfied and presumably in a stable state, so why the double bond need to “attack”? Does the double bond have a partial negative charge? I understand that pi bonds can break easily, but wouldn’t a force have to act upon the double bond in order for it to break and seek out the nucleophile?
Kate,
You ask a great question, one that I explain in the first video of this series. Be sure to watch it and let me know if you’re still confused