 When it comes to naming Aromatic compounds in organic chemistry, you will be required to know both common and IUPAC names. My Naming Aromatic Compounds videos teach you how to approach nomenclature. or CLICK HERE for my entire 21-video series on Naming Organic Compounds.
When it comes to naming Aromatic compounds in organic chemistry, you will be required to know both common and IUPAC names. My Naming Aromatic Compounds videos teach you how to approach nomenclature. or CLICK HERE for my entire 21-video series on Naming Organic Compounds.
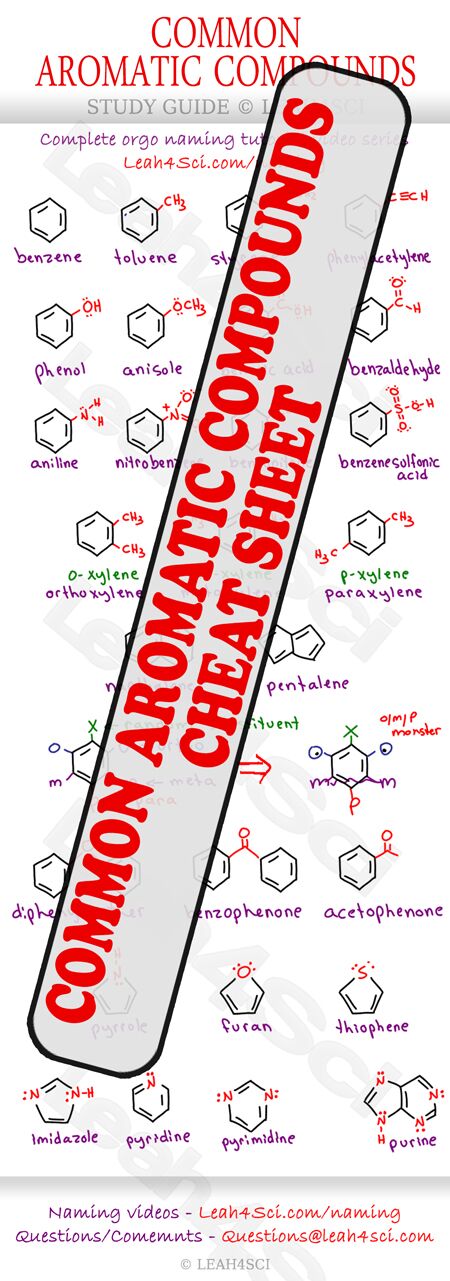
However, students often ask me for a list of ‘required' or ‘common' aromatic compounds. And so I've put together this detailed guide of various compounds you'll see in Organic Chemistry 1, 2, and perhaps even later on in Biochemistry/Biology.
This list is by no means complete, however it does cover most of what you'll see in organic chemistry.
This guide is free despite the hours I invested creating it. All I ask is that you share with your classmates by clicking on the Facebook/Twitter/Google+/Pinterest links above.
Right-click the image below then select ‘save as’ or print for the full version.



Hi..do you use WhatsApp? So i could get it
There…
Thank you very much. Your lectures are really helpful. You explain it in the best way.
Stay blessed. 🙂
Hellow.. Could you please help me, I tried to draw a structure for cyanide anion bound to BH3 but still my tutor told me it was wrong and I felt like giving up, It will be my pleasure if you help me. Thank you
I can not understand the aromatic heterocycles
Hi, Hareth. What specifically do you not understand about them?
How come Phenyl and Benzyl is not listed?
They are substituents, not molecules
Ma’am you are awesome..in love with organic chemistry..
Truly yours mam
Thank you
Thanks a lit for these cheat sheets. They’ve helped me a lot.
Please make one on aldehydes, ketones and amines.
You’re welcome Radhika. I don’t have plans to make another cheat sheet right now since I’m busy preparing for the Orgo Bootcamp later this month.
What is the orgo boot camp?
I’ll be discussing the bootcamp during this weekend’s workshops. Are you signed up?
You’re amazing. Thank you so much! 🙂
You’re very welcome Larissa
Can I ask you some questions about the substitution reaction of aromatic?
I´ve learnt that if there is a deactivating group on the aromatic ring; the alkylation and acylation rxn cannot occur. But if there also be an activating group on the ring, which one will determine the rxn? (Occur or not, position of the substituent
When there’s competition between activating and deactivating groups, the stronger activating group (if there’s more than one) wins. Be sure to take steric effects into consideration
Thankew you so much mam for this awesome work. It helded me alot and it will definitely tuned my knowledge to the proper stage…
You’re welcome Akshaya