 Intro to Orgo Series: Video 3
Intro to Orgo Series: Video 3
This tutorial video is designed to help new and incoming organic chemistry students build a solid foundation for organic chemistry
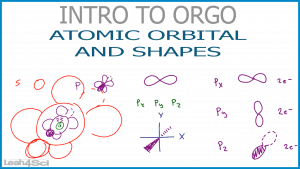
Topics discussed include an overview of the atomic orbitals including their shapes, sub-orbitals, and the number of electrons found in each. The orbitals are also related to their placement on the table leading into the topic of electron configurations.
(Watch on YouTube: Orbital & Shapes. Click cc on the bottom right for video transcription.)
Links & Resources Mentioned In This Video:
- Want to know more details about Orbitals? Check out my Quantum Numbers Mini Series.
<– Watch Previous Video: Periodic Table Trends
–> Watch Next Video: Electron Configuration Tutorial Part 1
This is Video 3 in the Intro to Orgo Video Series. Click HERE for the entire series.
Want to test your understanding? Try the Gen Chem Review for Orgo Quiz after watching the series!



You’r like lifejacket for my orgo
thanks from my heart
So is there more than one S orbital? S is the configuration, or shape, of the orbital, and 2s envelops 1s, 1p, 1f, and 1d?
There is the potential for ‘s’ at every principle energy level. Watch my quantum numbers video series to learn about possible orbitals per level https://leah4sci.com/mcat/mcat-chemistry/
why is electron not counted in mass.
why doesn,t it have weight
An electron weighs about 2000 times less than a proton, and so the weight is near zero in comparison
hahaha sooo funny…!!!! 😀 😀 😀
then wats d use of commenting??
I don’t understand your question