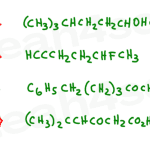Fischer projections crop in both organic chemistry and biochemistry. After studying my Fischer Projection tutorial series try your hand at this short quiz and see how you do. The questions range from simple to intermediate and are designed to help you figure out where you stand. If you find yourself stuck on any given problem […]
Cyclohexane Chair to Double Newman Quiz Challenge Question
This video walks you through the Chair Conformations Practice Quiz bonus question B – Converting a cyclohexane chair conformation to double Newman Projections. Practice is ESSENTIAL to mastering Organic Chemistry. You have to be able to apply the skills you are learning. First, complete the Cyclohexane Chair Conformations Practice Quiz, and then watch this video where I go over the Challenge Question […]
Stoichiometry and Reactions Practice Problems for MCAT Chemistry
While many students fear stoichiometry, I hope you’re feeling confident and ready to tackle practice problems after reviewing my MCAT Stoichiometry and Reactions video series. Haven’t watched it yet? Jump into the video series before attempting the quiz. This practice quiz was written to test your basic understanding of Stoichiometry and Reactions following along with AAMC Content […]
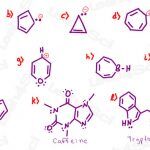
Aromaticity Practice Problems for Aromatic, Antiaromatic, Nonaromatic
Aromaticity is one of the more exciting topics in organic chemistry! Partly because of it’s potential for easy points on your quiz/exam.. that is IF you truly understand, not merely memorize: when you know what to look for to identify aromatic, antiaromatic and non aromatic compounds. Start with the Aromaticity Review + Videos, then see how […]
MCAT Style Non Calculator Math Practice Quiz
As an MCAT tutor I’m constantly telling my students to practice ‘simple math without a calculator’. The reply? Where can I find more practice problems? Since good resources are hard to find, I decided to write up my own MCAT math practice set NO Calculator Allowed! PDF Solution set at the end of the quiz […]
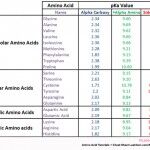
Amino Acids Practice Quiz
Amino acids are a big deal on the MCAT. You can’t simply ‘get’ the concept. You must be able to quickly recall information and apply them to questions. The quiz below will help you figure out if you’ve mastered this topic. The questions are simple but thorough. You either know it, or you don’t! Be sure […]
Resonance Practice Problems for Organic Chemistry
Think you know Resonance structures? Test your knowledge with my Resonance Structures Practice Quiz! Practice is ESSENTIAL to mastering Organic Chemistry. You have to be able to apply the skills you are learning. First, complete the Resonance Structures Practice Quiz, and then watch this video where I go over the first three question solutions and explanations step-by-step! (Watch […]
Acid Base Practice Problems Quiz
Ready to test your knowledge of acids and bases with a few practice problems? This quiz is designed to follow my Acids and Bases tutorial video series. But I’m not looking to see if you simply ‘memorized’ the concepts. The videos use simple examples to demonstrate concepts, these questions will test you at a slightly […]
Naming Organic Compounds Practice Problems with PDF Solutions
Naming organic compounds (aka IUPAC Nomenclature) will show up early in Organic Chemistry 1 and will follow you for the remainder of Orgo 1 AND Orgo 2. For example, when you’re asked to react [molecule name] with [molecule name], or to synthesize [molecule name]. If you’re here hoping to learn via practice but don’t yet […]
Oxidation Reduction Organic Chemistry Practice Quiz
Oxidation and Reduction reactions will come up over and over in your organic chemistry course. Think you’ve mastered redox reactions and reagents? Try the practice quiz below then scroll down (end of the quiz) for the PDF solutions. Not fully confident with redox? Review the redox tutorial video series and follow along with the Orgo Redox […]