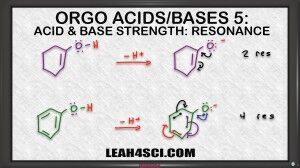
 Video 5 in the acid/base tutorial video series shows you how to use conjugate base resonance to determine the strength or stability of conjugate bases.
Video 5 in the acid/base tutorial video series shows you how to use conjugate base resonance to determine the strength or stability of conjugate bases.
If you can logically understand how resonance effects stability you'll have an easier time determining acidity of neutral starting acid molecules.
(click HERE to watch this video on YouTube. Catch video Transcript here)
Watch Previous Video: Ranking acids and bases A = Atom
Watch Next Video: Ranking acids and bases I = Inductive Effect
Ready to test your Acid/Base skills? Try my FREE acid/base practice quiz



Thanks a ton! This helped me more than a 30 minute long lecture about the same, in just 12 minutes. Again, thanks!
Hi Leah,
Isn’t there a 5th resonance structure to remove that charge off the last carbon? Are you not taking it into account because of its similarity to the first with oxygen holding the charge?
Thanks!
Thanks. I’m in general chem and this is helpful
Michelle: glad to help
Your videos are amazing! However, I have a doubt. We say that 1 electron carries -1 charge. But here when we transfer 2 electrons as a ‘lone pair’ we still transfer -1 charge. We should transfer -2 charge?
Sorry if my doubt is stupid, I’m just trying to learn.
One electron already belongs to the atom, the second electron is the one that introduces a negative charge
Hello! How are you? I want to appreciate to you.. you are doing good for all of us. you are leading me very well in chemistry. I am too much crazy to learn about chemistry. I hope you will help me more and more for my easiness.
Thank you, Muhammad!
thank you so much, your video help me so much
can you please make one about oxidation state of carbon atoms indifferent functional group, and oxidation level changes, and how we can differentiate if the reaction is an oxidation or reduction or neither
I’m sorry but I’m currently working on other video series
Your video are extremely helpful! Thank you so much for sharing.
You are very welcome Latonya. Are you in orgo 1 or orgo 2?