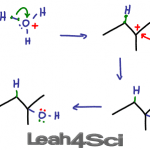
Acid catalyzed hydration of alkenes involves replacing the pi bond on an alkene with a water molecule. This is done by adding an alcohol to the more substituted carbon atom, and hydrogen to the less substituted carbon atom. This reaction follows Markovnikov’s rule and may undergo a carbocation rearrangement. If it is chiral, the product […]
Organic Chemistry Reference Material and Cheat Sheets

Alkene Reactions Overview Cheat Sheet – Organic Chemistry
The true key to successful mastery of alkene reactions lies in practice practice practice. However, … [Read More...]
MCAT Tutorials

Introduction To MCAT Math Without A Calculator
While the pre-2015 MCAT only tests you on science and verbal, you are still required to perform … [Read More...]
Organic Chemistry Tutorial Videos

Keto Enol Tautomerization Reaction and Mechanism
Keto Enol Tautomerization or KET, is an organic chemistry reaction in which ketone and enol … [Read More...]