The quiz below requires some mastery in reactions, intermediates, and step-by-step mechanisms. Not ready? Start by gaining your foundation in the Acetal Tutorial Video Series! Scroll to the very bottom of this quiz for a link to the FREE quiz solutions PDF (Coming Soon!) to ensure you’ve got this down. The solutions walkthrough video is available in the […]
Organic Chemistry Tutorial Videos
Halogenation Of Alkenes Tutorial Video
Alkene Reactions Series: Video 4 Halogenation of alkenes is the reaction in which a double bond is broken and replaced by a vicinal dihalide – 2 halogen atoms added to neighboring carbons. This reaction follows a pattern of anti addition. The goal of this video is to help you understand rather than memorize concepts related […]
Halohydrin Formation
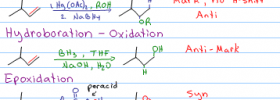
Alkene Reactions Series: Video 5 This tutorial takes you through the halohydrin formation mechanism, a type of electrophilic addition which adds both a halogen and alcohol to an alkene by using water instead of an inert solvent like CH2Cl2 or CCL4 to yield a halohydrin which features both a halogen and alcohol on the carbon […]
Acid Catalyzed Hydration of Alkenes
Acid catalyzed hydration of alkenes is the reaction in which a pi bond is broken and an alcohol added to the Markovnikov position when carried out in water. When alcohol is used as the reaction solvent, an ether will form following the same reaction mechanism. The video below is Part 6 in my Alkene Reaction […]
Hydride Shift and Methyl Shift Mechanism
Hydride shifts and methyl shifts can occur in organic chemistry reactions if there is a carbocation intermediate. While Markovnikov’s rule places the carbocation on the more substituted of the 2 former sp2 (double-bound) carbon atoms, if there is an EVEN MORE substituted carbon nearby, you’ll get a carbocation rearrangement. These videos help you understand the […]
Markovnikov vs Anti-Markovnikov in Alkene Addition Reactions
Markovnikov’s Rule tells us where to add the nucleophile and hydrogen in an asymmetrical alkene addition reaction. This is a critical pattern to both understand and recognize when studying alkene addition reactions. What is Markovnikov’s rule all about and how does this impact regioselectivity in electrophilic addition reactions? As you follow along with my alkene reactions […]
Naming Branched Substituents – Tert-Butyl, Isopropyl, isobutyl, tertbutyl, and more
Naming Organic Compounds Series: Video 4 This tutorial video shows you how to name organic compounds with substituents coming off their substituents. This includes the ‘long’ version of naming each individual group, and the accepted abbreviations like tert-butyl and isopropyl. (Watch on YouTube: Substituents. Click CC on bottom right for transcript.) Links & Resources Mentioned […]
Propyl Butyl and Pentyl Branched Substituents
When you hear n-butyl, secbutyl, isobutyl and tert butyl. Do these terms scare you? Learning Organic Chemistry is like learning a new language where certain prefixes and suffixes will mean different things. Different ways of arranging bits and pieces will bring you to different variations of compounds. But just like a new language, the more you see it, […]
R and S configuration for Lengthy Complex Substituents
Chirality/Stereochemistry Video Series: Video 5 R and S configurations are found by ranking substituents on a chiral carbon. This is relatively straight-forward when substituents contain 1-3 atoms. This gets more confusing when the substituents are long, complex, and contain many of the same atoms. This orgo tutorial video shows you how to approach lengthy and […]
Resonance Practice Problems for Organic Chemistry
Think you know Resonance structures? Test your knowledge with my Resonance Structures Practice Quiz! Practice is ESSENTIAL to mastering Organic Chemistry. You have to be able to apply the skills you are learning. First, complete the Resonance Structures Practice Quiz, and then watch this video where I go over the first three question solutions and explanations step-by-step! (Watch […]
- 1
- 2
- 3
- …
- 15
- Next Page »